Corrosion Corner
Accelerating corrosion
tests on spray packaging
Hello, everyone. It is often assumed that a higher storage
temperature will increase the rates of polymer and metal
corrosion and thus reduce the time needed for corrosion
testing. For example, it’s common to assume that 3–6 months of
testing at a high storage temperature will produce corrosion that
would be observed after several years of testing at room temperature.
However, routine use of high temperatures to shorten storage
test times often leads to unexpected corrosion.
Chemical reaction rates double for every 10°C increase in
temperature when the reaction has first order kinetics that are
controlled by the activation energy of the chemical reaction. However,
the corrosion rates of spray package materials do not follow
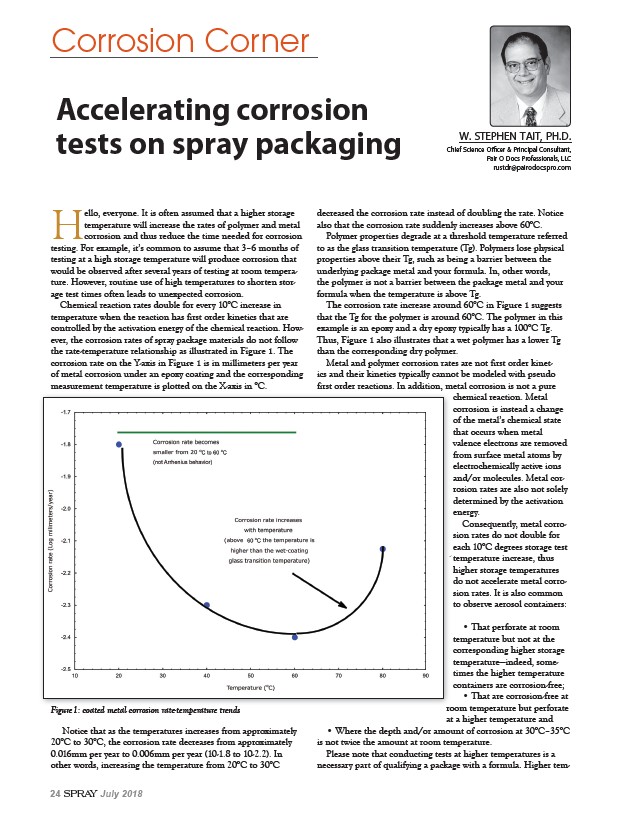
the rate-temperature relationship as illustrated in Figure 1. The
corrosion rate on the Y-axis in Figure 1 is in millimeters per year
of metal corrosion under an epoxy coating and the corresponding
measurement temperature is plotted on the X-axis in °C.
Figure 1: coated metal corrosion rate-temperature trends room temperature but perforate
Notice that as the temperatures increases from approximately
20°C to 30°C, the corrosion rate decreases from approximately
0.016mm per year to 0.006mm per year (10-1.8 to 10-2.2). In
other words, increasing the temperature from 20°C to 30°C
W. Stephen tait, ph.D.
Chief Science Officer & principal Consultant,
pair O Docs professionals, LLC
rustdr@pairodocspro.com
decreased the corrosion rate instead of doubling the rate. Notice
also that the corrosion rate suddenly increases above 60°C.
Polymer properties degrade at a threshold temperature referred
to as the glass transition temperature (Tg). Polymers lose physical
properties above their Tg, such as being a barrier between the
underlying package metal and your formula. In, other words,
the polymer is not a barrier between the package metal and your
formula when the temperature is above Tg.
The corrosion rate increase around 60°C in Figure 1 suggests
that the Tg for the polymer is around 60°C. The polymer in this
example is an epoxy and a dry epoxy typically has a 100°C Tg.
Thus, Figure 1 also illustrates that a wet polymer has a lower Tg
than the corresponding dry polymer.
Metal and polymer corrosion rates are not first order kinetics
and their kinetics typically cannot be modeled with pseudo
first order reactions. In addition, metal corrosion is not a pure
chemical reaction. Metal
corrosion is instead a change
of the metal’s chemical state
that occurs when metal
valence electrons are removed
from surface metal atoms by
electrochemically active ions
and/or molecules. Metal corrosion
rates are also not solely
determined by the activation
energy.
Consequently, metal corrosion
rates do not double for
each 10°C degrees storage test
temperature increase, thus
higher storage temperatures
do not accelerate metal corrosion
rates. It is also common
to observe aerosol containers:
• That perforate at room
temperature but not at the
corresponding higher storage
temperature—indeed, sometimes
the higher temperature
containers are corrosion-free;
• That are corrosion-free at
at a higher temperature and
• Where the depth and/or amount of corrosion at 30°C–35°C
is not twice the amount at room temperature.
Please note that conducting tests at higher temperatures is a
necessary part of qualifying a package with a formula. Higher tem-
24 Spray July 2018