W. Stephen Tait, Ph.D.
Chief Science Officer & Principal Consultant,
Pair O Docs Professionals, LLC
Hello, everyone. In the September edition of Corrosion Corner,
I began a five-part series on the corrosion behavior
of the various metals used to fabricate aerosol containers
and valves. This month I’ll discuss the corrosion behavior of
stainless steel, which is an iron alloy containing chromium and
nickel; the springs inside aerosol valves are typically made from
stainless steel.
Aerosol container valves
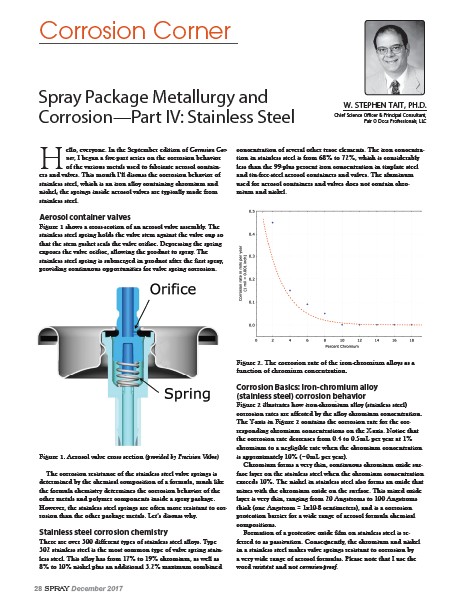
Figure 1 shows a cross-section of an aerosol valve assembly. The
stainless steel spring holds the valve stem against the valve cup so
that the stem gasket seals the valve orifice. Depressing the spring
exposes the valve orifice, allowing the product to spray. The
stainless steel spring is submerged in product after the first spray,
providing continuous opportunities for valve spring corrosion.
Figure 1. Aerosol valve cross section (provided by Precision Valve)
The corrosion resistance of the stainless steel valve springs is
determined by the chemical composition of a formula, much like
the formula chemistry determines the corrosion behavior of the
other metals and polymer components inside a spray package.
However, the stainless steel springs are often more resistant to corrosion
than the other package metals. Let’s discuss why.
Stainless steel corrosion chemistry
There are over 300 different types of stainless steel alloys. Type
302 stainless steel is the most common type of valve spring stainless
steel. This alloy has from 17% to 19% chromium, as well as
8% to 10% nickel plus an additional 3.2% maximum combined
28 Spray December 2017
concentration of several other trace elements. The iron concentration
in stainless steel is from 68% to 72%, which is considerably
less than the 99-plus percent iron concentration in tinplate steel
and tin-free-steel aerosol containers and valves. The aluminum
used for aerosol containers and valves does not contain chromium
and nickel.
Figure 2. The corrosion rate of the iron-chromium alloys as a
function of chromium concentration.
Corrosion Basics: Iron-chromium alloy
(stainless steel) corrosion behavior
Figure 2 illustrates how iron-chromium alloy (stainless steel)
corrosion rates are affected by the alloy chromium concentration.
The Y-axis in Figure 2 contains the corrosion rate for the corresponding
chromium concentrations on the X-axis. Notice that
the corrosion rate decreases from 0.4 to 0.5mL per year at 1%
chromium to a negligible rate when the chromium concentration
is approximately 10% (~0mL per year).
Chromium forms a very thin, continuous chromium oxide surface
layer on the stainless steel when the chromium concentration
exceeds 10%. The nickel in stainless steel also forms an oxide that
mixes with the chromium oxide on the surface. This mixed oxide
layer is very thin, ranging from 20 Angstroms to 100 Angstroms
thick (one Angstrom = 1x10-8 centimeters), and is a corrosion
protection barrier for a wide range of aerosol formula chemical
compositions.
Formation of a protective oxide film on stainless steel is referred
to as passivation. Consequently, the chromium and nickel
in a stainless steel makes valve springs resistant to corrosion by
a very wide range of aerosol formulas. Please note that I use the
word resistant and not corrosion-proof.
Corrosion Corner
Spray Package Metallurgy and
Corrosion—Part IV: Stainless Steel